We are witnessing a death march throughout the world for last 5 months. According to the reliable WORLDOMETER estimates, latest (12.05.2020) worldwide figure of Covid-19 or SARS-COV-2 shows – coronovairus cases 4,277,258, deaths: 287,698, recovered: 1,537,7646. Most ruefully, it is an unusual and dreaded situation where people cannot jump into the situations like fire, earth quake, flood or famine where every sensible person extends his/her hands to get hold of the person in distress. Contrarily, here personal and social distancing are the key words to remain safe and save our social existence. In a paper “Profile of a killer virus”, published in Nature (7.05.2020), it has been found – “A neighbour’s cough that sends ten viral particles your way might be enough to start an infection in your throat, but the hair-like cilia found there are likely to do their job and clear the invaders. If the neighbour is closer and coughs 100 particles towards you, the virus might be able get all the way down to the lungs.” As we know till date quite little about what precise pathophysiological, biochemical, haematological and other changes the coronavirus cause within our human body system and how it remains shrouded in clouds of unknowingness and uncertainty. The Nature paper further observes – “it might work its way down to the lungs and debilitate that organ. How it gets down there, whether it moves cell by cell or somehow gets washed down, is not known.” Such is the condition of our present knowledge – we are not much sure about how the virus exactly moves from the respiratory tract to the lungs. As a result, there remains groping for definite therapeutic strategies.
In the editorial “Covid-19 – Navigating the Uncharted” (26.03.2020), published in New England Journal of Medicine (NEJM), it is commented – “Therapy currently consists of supportive care while a variety of investigational approaches are being explored. Among these are the antiviral medication lopinavir–ritonavir, interferon-1β, the RNA polymerase inhibitor remdesivir, chloroquine, and a variety of traditional Chinese medicine products. Once available, intravenous hyperimmune globulin from recovered persons and monoclonal antibodies may be attractive candidates to study in early intervention. Critical to moving the field forward, even in the context of an outbreak, is ensuring that investigational products are evaluated in scientifically and ethically sound studies.”
No definite answer is before us. What we lack is a specific antiviral agent to treat the infected and, optimally, decrease viral shedding and subsequent transmission. Presently much uproar is there around remdesivir – an investigational drug. We will have to wait further for more double-blind, placebo-controlled, randomized controlled trials of this drug to say finally how much hype it can sustain.
I have randomly selected a few countries to show the number of tests they have done per million population. These are like these – US: 29,063 / UK: 28,309 / Italy: 43,112 / Spain: 52,781 / France: 21,213 / Germany: 32,891 / Iran: 7,328 / Denmark: 57,709 / South Korea: 13,281 / Australia: 34,429 / New Zealand: 40,870 / Israel: 54,277 / Thailand: 3,260 / Vietnam: 2,681 / India: 1,275 / Singapore: 30,016 / Bangladesh: 830 / Pakistan: 1,385 / China: Figure unvaialable / Sri Lanka: 1,759.
Here, it is important to remember Vietnam has got 0 (zero) death despite its fragile economy, health care with little resource and 1,100 kms land border with China. How did this miracle happen? We will see later on. New Zealand has reported 0 deaths for last 5 days. Australia is nearing towards zero. Contrarily, new infections and re-infections are being reported in Singapore, South Korea and China. As evident from the figures above the lesser the tests and “contact tracing” per million population the more will be the number of “silent super-spreaders” in a society, who are potentially fatal once lockdown is lifted and people are allowed for social mixing in any population-dense country as in India or Bangladesh. It can be ominous for us.
US alone crosses the mark of 81,000 deaths – more than official deaths in Vietnam war. Importantly, first infection cluster reported in Wuhan since lockdown was lifted. Iran orders lockdown anew for southwest country after spike in infection. I shall deal further with these issues later on.
VIRUS BIOLOGY – They can be stored for several years at -80*C and can be inactivated at 56*C for 30 minutes.
A virus is “simply a piece of bad news wrapped up in protein,” the biologists Jean and Peter Medawar wrote in 1977.
Potential for mutations/one RNA thread
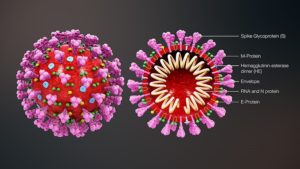
Truly speaking, coornavirus is a “genetic instruction” which is enveloped by oily substance. The genome of the new coronavirus is less than 30,000 “letters” long. (The human genome is over 3 billion.) Scientists have identified genes for as many as 29 proteins, which carry out a range of jobs from making copies of the coronavirus to suppressing the body’s immune responses to producing plethora of symptoms including fever, cough, severe respiratory distress, loss of appetite and smell, blood clotting disorders leading to young-age strokes, acute myocardial infarctions and many more.
Beginning of RNA thread^ 30,000 letters Spike protein End of RNA
Viruses must hijack living cells to replicate and spread. When the coronavirus finds a suitable cell, it injects a strand of RNA that contains the entire coronavirus genome. One recent study shows that Coronavirus has mutated into 10 different types. The coronavirus can be classified into many types — O, A2, A2a, A3, B, B1, and so on
Currently, there are 11 types, including type O which is the ‘ancestral type’ that originated in Wuhan. The A2a coronavirus is highly efficient in entering the lung cells in large numbers. The A2a is hence easily transferable resulting in the rapid spread of the coronavirus across the world, the authors of the report wrote.
The first viral protein created inside the infected cell is actually a chain of 16 proteins joined together. Two of these proteins act like scissors, snipping the links between the different proteins and freeing them to do their jobs.
The virus contains Spike Proteins to anchor to ACE2 receptors of human lungs. There are 2 other non-structural proteins – NSP1 and NSP2.
Cellular Saboteur NSP1 – This protein slows down the infected cell’s production of its own proteins. This sabotage forces the cell to make more virus proteins and prevents it from assembling antiviral proteins that could stop the virus.
Mystery Protein NSP2 – Scientists aren’t sure what NSP2 does. The other proteins it attaches to may offer some clues. Two of them help move molecule-filled bubbles called endosomes around the cell.
The coronavirus binds to host cells and fuses with the cell’s membrane, releasing its viral genome and hijacking the cell’s machinery to duplicate its RNA and create new viral proteins. The RNA and proteins are packaged into new virus particles, which are excreted from the cell to infect more cells.
One paper “Receptor Recognition by the Novel Coronavirus from Wuhan: an Analysis Based on Decade-Long Structural Studies of SARS Coronavirus” published in the Journal of Virology (April, 2020) comments – “Our decade-long structural studies on the receptor recognition by SARS-CoV have identified key interactions between SARS-CoV spike protein and its host receptor angiotensinconverting enzyme 2 (ACE2), which regulate both the cross-species and human-to-human transmissions of SARS-CoV.”
The SARS-CoV-2 genome also encodes a group of so-called “accessory proteins.” They help change the environment inside the infected cell to make it easier for the virus to replicate. In a paper – “Phylogenetic network analysis of SARS-COV-3 genomes” – researchers find – “In a phylogenetic network analysis of 160 complete human severe acute respiratory syndrome coronavirus 2 (SARS-Cov-2) genomes, we find three central variants distinguished by amino acid changes, which we have named A, B, and C, with A being the ancestral type according to the bat outgroup coronavirus. The A and C types are found in significant proportions outside East Asia, that is, in Europeans and Americans. In contrast, the B type is the most common type in East Asia, and its ancestral genome appears not to have spread outside East Asia without first mutating into derived B types, pointing to founder effects or immunological or environmental resistance against this type outside Asia.”
From zoonotic to anthropozoonotic – For thousands of years, a parasite with no name lived happily among horseshoe bats in southern China. The bats had evolved to the point that they did not notice; they went about their nightly flights unbothered. One day, the parasite—an ancestor of the coronavirus, SARS-CoV-2—had an opportunity to expand its realm. Perhaps it was a pangolin. In a paper published in Nature (“The proximal origin of SARS-CoV-2” – 17.03.2020) it was observed – “the high-affinity binding of the SARS-CoV-2 spike protein to human ACE2 is most likely the result of natural selection on a human or human-like ACE2 that permits another optimal binding solution to arise. This is strong evidence that SARS-CoV-2 is not the product of purposeful manipulation.”
Another paper “Escaping Pandora’s Box — Another Novel Coronavirus” in NEJM (2.04.2020) categorically pointed out – “We must realize that in our crowded world of 7.8 billion people, a combination of altered human behaviors, environmental changes, and inadequate global public health mechanisms now easily turn obscure animal viruses into existential human threats.”
It is human greed for maximization of profit and commoditization of everything and everyone that has made possible the virus happily harbouring within bats for millions of years for spillover to human body in a very short span of time. A research paper (“Anthropogenic Land Use Change and Infectious Diseases: A Review of the Evidence” – 23.05.2014) published in the journal EcoHealth finds – “Land use change has the potential to impact disease dynamics directly and indirectly by changing the abundance, demography, behaviour, movement, immune response, and contact between host species and vectors, as well as altering host community composition.” Our mastery over nature has bounced back.
The virus harbouring within bats for millennia has spilled over to human habitation. Human body is their new hostage. Why? Corporate greed and avarice for ever-increasing highest profit has devastated human being-ecology-nature-living world balance and many viruses have lost their natural habitat- now encroaching on to human world.
But it was not supposed to be. After the SARS-COV-1 (2002-3) and MERS pandemic PREDICT project was initiated by USAID’s of Emerging Pandemic Threats in 2009. It was aimed to enable global surveillance of pathogens that can spillover form animal hosts to people by building capacities to detect and discover viruses within several important virus groups such as coronavirus. According to Los Angeles Times (2.042020), just at the beginning of the Wuhan reports “Trump administration ended pandemic early-warning programs to detect coronaviruses”. Moreover, ““the Trump administration ended a $200-million pandemic early-warning program aimed at training scientists in China and other countries to detect and respond to such a threat.” When the program was stopped it had already detected and discovered 1,200 various types of viruses amongst which 160 were coronaviruses. Around the globe, including Wuhan, scientists and technicians were being trained in 60 different laboratories to enhance the project.
The closure of the PREDICT project did irreparable harm to science and, also, to humankind. Science yielded to politics and state.
Weapons in our arsenal in fighting against the coronavirus – We know very little about this virus, especially pathophysiological cascades which lead to the conglomerate of symptoms, and, finally, the two deadly outcomes – (a) cytokine storm syndrome, and (b) fulminant myocarditis, which almost invariably lead to death. Before dealing with therapeutic in an elaborate way, let me take a pause and discuss about some latest research findings regarding the virus.
CDC has described 6 new symptoms (along with usually known symptoms) which may appear 2-4 days after exposure to the virus – mean period being 5 days. A Covid-19 patient may have combination of symptoms. To mention, children have similar symptoms to adults and generally have mild illness. Symptoms include –
- Cough
- Shortness of breath or difficulty breathing
OR at least 2 of these symptoms
- Fever
- Chills
- Repeated shaking with chills
- Headache
- Sore throat
- New loss of taste or smell
Newer studies show significant number of strokes in patients of age less than 50, heart attack, and disseminated intravascular coagulation (DIC), along with other haematological disorders and neurological problems. NEJM reports (28.04.2020) “Large-Vessel Stroke as a Presenting Feature of Covid-19 in the Young”.
To date, 80-81% of patients have milder symptoms and recover completely, 14% require hospitalization, and 5% need ICU, ventilators, hyperbaric oxygenation and other trial-and-error therapeutic molecules.
In no other journal than Nature Medicine (15.04.2020) there are intriguing findings regarding viral shedding and transmissibility of COVID-19 (“Temporal dynamics in viral shedding and transmissibility of COVID-19”) – “We observed the highest viral load in throat swabs at the time of symptom onset, and inferred that infectiousness peaked on or before symptom onset. We estimated that 44% (95% confidence interval, 25–69%) of secondary cases were infected during the index cases’ presymptomatic stage, in settings with substantial household clustering, active case finding and quarantine outside the home. Disease control measures should be adjusted to account for probable substantial presymptomatic transmission.” Researchers collected samples from 30 different places and analyzed viral loads in different areas and possible infectivity.
- Patient areas: Viral RNA concentrations generally were very low or undetectable in patient areas (e.g., ICUs, coronary care unit), except in a patient mobile toilet room, which was not ventilated.
- Medical staff areas: Some sites — including rooms where personal protective equipment was removed — had high SARS-CoV-2 RNA levels; these levels became undetectable after better sanitization procedures were implemented.
- Public areas: Two areas that got a lot of foot traffic — the entrance to a department store and a site next to one of the hospitals — had high viral RNA concentrations.
In a paper – “Covert coronavirus infections could be seeding new outbreaks” – in Nature (20.03.2020) it was observed – “As coronavirus outbreaks surge worldwide, research teams are racing to understand a crucial epidemiological puzzle — what proportion of infected people have mild or no symptoms and might be passing the virus on to others. Some of the first detailed estimates of these covert cases suggest that they could represent some 60% of all infections.” In a more recent editorial (“Asymptomatic Transmission, the Achilles’ Heel of Current Strategies to Control Covid-19”) in NEJM (27.04.2020) it is commented – “Quantitative SARS-CoV-2 viral loads were similarly high in the four symptom groups (residents with typical symptoms, those with atypical symptoms, those who were presymptomatic, and those who remained asymptomatic). It is notable that 17 of 24 specimens (71%) from presymptomatic persons had viable virus by culture 1 to 6 days before the development of symptoms. Finally, the mortality from Covid-19 in this facility was high; of 57 residents who tested positive, 15 (26%) died.”
Analyzing these studies in the most recent paper (29.04.2020) in the Lancet it is suggested that “Institutional, not home-based isolation could contain the COVID-19 outbreak”.
Therapeutic Arsenal – Despite Trump’s touting hydroxycholoroquine and azithromycin as “game changers”, not a single, large, double-blind, randomized controlled trial (RCT) has proven their efficacy, rather the reverse one – very bad prognosis. We do not have any definitive therapy or preventing measure (excepting hand washing, cough etiquette, frequent cleansing, social distancing of 6 feet etc.) for the virus. But Trump’s touting of HCQ and Azithromycin as “game changers” and almost instant approval of the drugs under EUA (Emergency Use Authorization) from an autonomous, dignified and acceptable organization like FDA and, later NIH (National Institute of Health), further leads us to believe in the medical-state-politics-industrial complex.
JAMA (Journal of American Medical Association) had their observation in “Pharmacologic Treatments for Coronavirus disease-2019 (COVID-10)” – “Currently, there is no evidence from randomized clinical trials (RCTs) that any potential therapy improves outcomes in patients with either suspected or confirmed COVID-19. There are no clinical trial data supporting any prophylactic therapy. More than 300 active clinical “treatment trials are underway.” NEJM commented – “A robust research effort is currently under way to develop a vaccine against Covid-19. We anticipate that the first candidates will enter phase 1 trials by early spring. Therapy currently consists of supportive care while a variety of investigational approaches are being explored. Among these are the antiviral medication lopinavir–ritonavir, interferon-1β, the RNA polymerase inhibitor remdesivir, chloroquine, and a variety of traditional Chinese medicine products.” (Editorial – “Covid-19 – Navigating the Uncharted” – 26.03.2020)
It simply means there is not a single positive therapy for the virus so far.
The Question of Immunity – WHO declared in a statement ob 24th April, 2020, that there is no “IMMUNITY PASSPORTS” in the sense that there is currently no evidence that people who have recovered from COVID-19 and have antibodies are protected from a second infection. Such things are occurring in South Korea, Singapore and, more recently in Wuhan, China. There is not enough antibody in the patients recovering from Covid-19 infection and they are getting re-infected. As of 24 April 2020, no study has evaluated whether the presence of antibodies to SARS-CoV-2 confers immunity to subsequent infection by this virus in humans.
People who assume that they are immune to a second infection because they have received a positive test result may ignore public health advice. The use of such certificates may therefore increase the risks of continued transmission. As new evidence becomes available, WHO will update this scientific brief.
The question of herd immunity cannot also be thought of right at this moment. In this hour great human crisis and uncertainty people become credulous. They tend to be more emotional to belive in rumours and pseudoscience than in reason, logic and rationality.
Science, Reason vs. Emotion and Pseudoscience
Nature in a report (27.04.2020) “Pseudoscience and COVID-19 — we’ve had enough already” has warned its readers – “Cow urine, bleach and cocaine have all been recommended as COVID-19 cures — all guff. The pandemic has been cast as a leaked bioweapon, a byproduct of 5G wireless technology and a political hoax — all poppycock. And countless wellness gurus and alternative-medicine practitioners have pushed unproven potions, pills and practices as ways to ‘boost’ the immune system.” In its further observation – “Thankfully, this explosion of misinformation — or, as the World Health Organization has called it, the “infodemic” — has triggered an army of fact-checkers and debunkers. Regulators have taken aggressive steps to hold marketers of unproven therapies to account. Funders are supporting researchers (myself included) to explore how best to counter the spread of COVID-19 claptrap.
NEJM (April 29, 2020) has also warned us about this necessity in “Covid-19 – A Reminder to Reason” – “In a time when the rational–emotional scale is tipping to the emotional side, we begin relying more heavily on anecdotes, particularly personal experiences that may carry inordinate weight in our minds. Journalists use the power of stories to connect with readers and tug at their emotions. Physicians, trained as scientists, are expected to follow a hypothesis-driven, rational, evidence- based approach to clinical decision making, but we, too, can be swayed by stories under the pressures of a crisis.” More importantly, the thirst for a treatment in an uncertain time led to flawed studies that may have contributed to negative physical, social, and economic consequences.
Definite Therapeutic Interventions – Right at this moment we have two armaments to treat prospective and ongoing Covid-19 patients. First, remdesivir and, second, vaccine.
First trial of remdesivir was conducted in 61 hospitalized patients and the report was published in NEJM on April 10, 2020, under the title “Compassionate Use of Remdesivir for Patients with Severe Covid-19”. The report showed – During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 of 30 patients (57%) receiving mechanical ventilation who were extubated. A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
Finally, on April 29, 2020, pharma giant Gilead issued a news release saying it had become aware of results from the NIAID trial showing its experimental drug remdesivir helped patients recover more quickly than standard care, suggesting it could become the first effective treatment for an illness that has turned modern life inside-out. NIAID’s trial enrolled more than 1,000 patients internationally and compared remdesivir treatment alongside supportive care with a placebo. Patients who got the drug recovered in an average of 11 days, while those who get a placebo recovered in 15 days.
It has been approved by FDA only for hospital use, in EUA (Emergency Use Authorization) category till date. Interestingly, Gilead shares jumped as much as 8.1% Wednesday after the company’s statement. Remdesivir is consistent with Trump’s electoral politics of America First slogan. Wall Street Journal (1.05.2020) reports “Remdesivir Affirms the American Way”. NPR news agency (2.05.2020) reports – “Gilead Lobbying Rose As Interest in COVID-19 Treatment Climbed … The pharmaceutical company spent $2.45 million on lobbying in the first three months of the year, a 32% increase over the $1.86 million it spent in the first quarter of 2019.”. POGO (Project on Government Oversight) published reports (1.01.2026) on FDA in two instalments – (1) FDA Depends on Industry Funding; Money Comes with “Strings Attached”, and (2) DRUG MONEY: In FDA Meetings, ‘Voice’ of the Patient Often Funded by Drug Companies.
In a research paper – “Remdesivir in adults with severe COVID-19: a randomized, double-blind, placebo-controlled, multicentre trial” – published on April 29, 2020, in Lancet does express some reserved observation. To note, this trial is double-blind and placebo-controlled, while that conducted by Gilead and NIH is open-label.
According to the Lancet paper – “We did a randomised, double-blind, placebo-controlled, multicentre trial at ten hospitals in Hubei, China. Eligible patients were adults (aged ≥18 years) admitted to hospital with laboratory-confirmed SARS-CoV-2 infection, with an interval from symptom onset to enrolment of 12 days or less, oxygen saturation of 94% or less on room air or a ratio of arterial oxygen partial pressure to fractional inspired oxygen of 300 mm Hg or less, and radiologically confirmed pneumonia. Patients were randomly assigned in a 2:1 ratio to intravenous remdesivir (200 mg on day 1 followed by 100 mg on days 2–10 in single daily infusions) or the same volume of placebo infusions for 10 days. Patients were permitted concomitant use of lopinavir–ritonavir, interferons, and corticosteroids … In this study of adult patients admitted to hospital for severe COVID-19, remdesivir was not associated with statistically significant clinical benefits. However, the numerical reduction in time to clinical improvement in those treated earlier requires confirmation in larger studies.”
Gilead Sciences, the “discoverer” of this failed drug so far, publishes on its own website (7.05.2020) – Due to the current public health emergency, the U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization for remdesivir for the treatment of COVID-19. In the United States, remdesivir is an investigational drug that has not been approved by the FDA for any use, and the safety and efficacy of remdesivir for the treatment of COVID-19 has not been established. The distribution of remdesivir in the United States has been authorized only for the treatment of hospitalized patients with severe COVID-19 … Remdesivir is not yet licensed or approved outside of Japan and ongoing clinical trials continue to evaluate its safety and efficacy.
Do we need to comment further? It is superfluous.
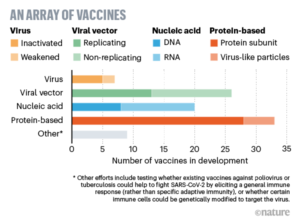
Vaccine – first important paper was published in the journal EBioMedicine on April 2, 2020 – Microneedle array delivered recombinant coronavirus vaccines: Immunogenicity and rapid translational development. MNA (micro-needle array) delivered MERS-S1 subunit vaccines induced potent and long-lasting antigen antigen-specific immune responses. Notably, MNA delivery of these vaccines generated significantly stronger immune responses than those administered by traditional subcutaneous needle injection, indicating the improved immunogenicity by skin-targeted delivery. MNA-enabled vaccine has two important properties – (1) MNA-embedded vaccines have the potential to remain stable for an extended period of time without expensive “cold chain” requirements, and (2) previous animal and clinical studies suggest that MNAs could provide a safe and well-tolerated delivery platform for efficacious immunization strategies. Moreover, vaccine produced in this method is not inactivated after gamma-ray sterilization. According to the paper – “Combining emerging biotechnology methods with bioengineering advances in vaccine delivery strategies, it may now be possible to rapidly produce clinically-translatable vaccines against novel pathogens for human testing and subsequent global distribution in time to significantly impact the spread of disease.
Right now, more than 90 MNCs and academia are in breakneck competition for vaccine development. Phase 1 human has already been started. Hundreds of people volunteer to be infected with coronavirus. Most likely, we will have to wait for at least 8 months more for the vaccine to come in the market. Even then, it is very unlikely that it will be universally available owing to both price constraint and production limit. About two months ago (18.03.2020) Nature asked 5 key questions (“Coronoavirus vaccines: five key questions as trials begin”) – (1) Do people develop immunity? (2) If humans do develop immunity, how long does it last? (3) What kind of immune response should vaccine developers look for? (4) How do we know if a vaccine is likely to work? And (5) Will it be safe?
Coronavirus can change the paradigm of comprehensive health care-based system forever. It will possibly lead to disease-centred vertical programs – hi-tech with ventilators, ECMO, costly drugs and other technologies. Also, it may lead personal distancing between the patient and the doctor. Telemedicine and virtual treatments may fill in the gaps.
Finally, we have to be always cautious with –
- hand washing frequently or alcohol-based sanitizing
- Cough and sneezing etiquette
- Use of face mask of proper quality
- Personal and social distancing of 6 feet.
Our world will never be the same after Covid-19 pandemic. We will have to be prepared for other pandemics to come. We will also have to learn to live with the virus which has come out of the Pandora’s Box of nature devastated by us.
As we push ahead, we should take heart in the Hesiod version of the Pandora myth, in which Pandora managed to prevent a single escape: “Only Hope was left . . ., she remained under the lip of the jar, and did not fly away.” (“Escaping Pandora’s Box — Another Novel Coronavirus”)










Very important article.Beautifully articulated. Thanks,Dr.
Very interesting article, very comprehensive. The point to note is that despite little we know about the virus, traditional public health practices still seem to offer the best evidence based results. This is evident in almost everywhere the system went with a strong public health response with washing hands, distance maintenance, etc.
There is no real evidence based treatment that has shown to have a high enough benefit:risk ratio to be clinically usable.
Your speculation about how this is going to change medical practice and culture is very interesting. A space to watch.
Excellent article, answered lot of unanswered questions.
Very nice.
Read your interesting and enlightening article / articles with absorbing interest about Corona virus and the mystery in which it is shrouded. You have done a praiseworthy job of making available to the lay reader like me the gist of so many scholarly and well-resrarched articles published in famous medical journals of the world. Congratulation. Subhas Chandra DASGUPTA. Dansiri,Collegepara, Raiganj.
Thank you all respondents!
Like!! Great article post.Really thank you! Really Cool.
I learn something new and challenging on blogs I stumbleupon everyday.
I really like and appreciate your blog post.
Worth-reading article with extensive, rational, timely discussion to enlighten the laymen like ours and put the ever-chirping little-learners in adequate light.
Yet another masterpiece from none other than Dr Jayanta Bhattacharya who has constantly empowered us with the necessary knowledge during these difficult times. I must thank Dr JB and Doctors Dialogue for their huge efforts.
Thank you all, especially Kanchan and Bhaskar!
Only a person who has assimilated all the facts properly and scientifically can represeed them in a lucid fashion for common people. Mark of a true academician. However, the comment on the changed paradigm of the health care system makes me nervous. Can we do it for everybody? Regards Dr. Bhattacharya.
Thank you Buddhadeb Bagchi. It’s a concerted collective effort to make a journey towards a better future.
অনেক উচ্চমানের তথ্য সমৃদ্ধ প্রবন্ধ পেশ করেছেন ডঃ জয়ন্ত ভট্টাচার্য। বেশ কিছু আলোচনা আমার মত অবৈজ্ঞানিকের আয়ত্তের বাইরে।
তবুও মূল বিষয়টি বোধহয় বুঝতে পেরেছি।
কোভিড সহজে যাবে না। নব নব রূপে আসতেই থাকবে। ভাইরাসের চরিত্র সম্পূর্ণ বিশ্লেষণ এখনও হয় নি, অতএব চালু প্রতিষেধকগুলো পুরো কাজ করতে পারবে না। আক্রান্তের রোগ নিরাময়েরও কোন সুনির্দিষ্ট ওষুধ এখনও নেই। সুতরাং স্বাস্থ্যবিধি মানলেই বাঁচোয়া, নইলে নয়।
ধন্যবাদ ডঃ ভট্টাচার্য আমাদের সচেতন করার জন্য।